MCHFT is a research active Trust.
For more information on specific research areas, click on the links below.
Research is actively promoted in our Paediatric and Neonatal areas and we have a wide range of studies covering various specialties from Respiratory, Orthopaedics, Dermatology and Endocrinology.
If you would like to find out more, please call 01270 612123.
What is a Clinical Trial? - a short video for parents and children
https://
Open Trials

The BADBIR research study is an observational study looking at treatments used for patients with psoriasis.

The ADDRESS-2 project is for children and adults newly diagnosed with type 1 diabetes wanting to hear about opportunities to take part in clinical research.

GenOMICC studies the genes (DNA) of people who become critically ill, or meet certain criteria, to better understand why some people become sicker than others and, potentially, to discover new ways of treating patients. More info can be found at www.

Cytomegalovirus, or CMV, is a common virus that can infect people of all ages. CMV can pose serious problems to unborn babies if a woman catches the virus in pregnancy. The aim of the study is to gather information about children born with CCMV in the UK, including identifying risk factors for children who have long term problems, such as hearing loss, and to see which tests may be able to predict these problems early on. We will also monitor the side effects of treatment. We hope this study will support the future development of new strategies and therapies for better treatment and prevention of CCMV.

B-STOP- Biomarkers of Systematic Treatment Outcomes in Psoriasis
This study can only be offered to patients who are participating in the BADBIR study. The overall research aim of this large, multi-centre study is to discover which gene(s) and other so- called ‘biomarkers’ (for example present in blood sample) are important in determining good (or poor) responses to the drugs that are used to treat psoriasis.

Bronchstart is a national study looking children who have Bronchiolitis. This is a very common winter condition caused by viral infections. The study has been running for 3 years and initially looked at the impact of COVID-19 on bronchiolitis. This year we are examining how a vaccine being given to mothers helps stops children being admitted with Bronchiolitis.
If you require any more information, please contact joanne.
Trials no longer recruiting but still collecting follow-up data:
Aztec 2
CFSTART
CRAFFT
BLIPA
Leighton Hospital’s Surgery and Cancer Research Department is actively involved in clinical research and has many studies currently open to recruitment across seven different disease areas.
If you would like to know more about getting involved in any clinical research in any of these areas please contact the Surgery and Cancer Research Department on 01270 273486 or email oncologyandsurgeryresearch@mcht.nhs.uk
OPEN TRIALS
BREAST

A clinical study for people with early stage breast cancer which has spread to the axillary nodes. Breast cancer sometimes spreads to other areas of the body via the lymphatic system. Chemotherapy can be given as a first treatment to target these cells and reduce cancer in the axillary before surgery. After chemotherapy , further treatment to the axillary is usually offered to everyone which may include surgery or radiotherapy. This extra treatment can cause additional side effects like lymphedema and shoulder problems. We are investigating whether the extra axillary treatment is necessary for all patients to ensure we offer patients the best treatment with the fewest side effects.

Case control study to identify the best approach to breast cancer risk assessment in women in their 30s and to assess its acceptability.

Open Surgery versus Minimally invasive vacuum-Assisted excision for smaLL screen-detected breast cancer – a phase III randomised multi-centre trial.
COLORECTAL

FOXTROT2 - currently paused to recruitment
A phase III trial of neo-adjuvant chemotherapy in older and/or frail patients with locally advanced but resectable colon cancer.
To assess and compare whether 6 weeks of NAC with oxaliplatin/fluoropyrimidine (OxFp) followed by surgery will improve disease free survival (DFS) and short-term safety and efficacy outcomes in frail and/or older patients compared with straight to surgery (STS). All participants will be followed up pre & post NAC and pre & post-surgery. Recurrence events will be collected on annual follow up reports.

FOXTROT3 - currently paused to recruitment
A randomized phase III trial of neo-adjuvant chemotherapy with mFOLFOXIRI compared with neoadjuvant OxFp in patients with locally advanced but resectable colon cancer. To assess and compare whether 6 weeks of NAC with mFOLFOXIRI followed by surgery will improve the proportion of patients with a pathological response to NAC (as assessed in the surgical resection sample) according to tumour regression grade (TRG), disease-free survival (DFS) and short-term safety and efficacy outcomes compared with 6 weeks of NAC with OxFp followed by surgery. All participants will be followed up pre & post NAC and pre & post-surgery. Recurrence events will be collected on annual follow up reports.
TRACC

Tracking mutations in cell free tumour DNA to predict Relapse in eArly Colorectal Cancer. To demonstrate a de-escalation strategy of ctDNA guided adjuvant chemotherapy is non-inferior to standard of care treatment as measured by 3 year disease free survival in patients with high risk stage II or stage III colorectal cancer with no evidence of minimal residual disease (ctDNA negative).
SURGICAL

“Perioperative” refers to the time before, during and after surgery. Our aim is to improve the care and treatment of patients undergoing major surgery in the United Kingdom. We do this by collecting and studying information about you, your surgery, and then your recovery afterwards.
ORTHOPAEDIC

This study is looking at the effectiveness of total knee replacement with Patellar Resurfacing.

The HIPPY study aims to find out which type of hip implant currently available on the NHS is best for patients in the long term, and most cost effective for the NHS.
THYROID

A randomised control trial to compare thyroid cancer recurrence, quality of life, surgical morbidities/effects, and cost-effectiveness between total thyroidectomy and hemithyroidectomy (HT) in a national cancer setting.
LUNG

TOURIST trial is a Phase III study aimed at evaluating the efficacy of thoracic radiotherapy in treating stage IV non-small cell lung cancer (NSCLC) using advanced radiotherapy techniques.
Trials no longer recruiting but still collecting follow-up data:
MAMMO-50 (Observational non RCT)
MAMMO-50
PATCH
Persephone
PRIME II
MARVEL
POETIC
IMPRESS
POSNOC
B-AHEAD3
True NTH UK Post Surgical Follow up
PRIMETIME
ESPAC-4
E-PATH
BOSS
SUPREMO
NSCCG
IMPORT HIGH
ACE
BEST BRA
STAMPEDE
ADD-ASPIRIN
OPTIMA
POPPY
ROSSINI
THE UKGPCS
CITRUS
Multiple Pregnancy Register
To systematically track complicated multiple pregnancies, and uncomplicated monochorionic twin pregnancies, to ascertain pregnancy outcomes and address the gaps in knowledge.
To improve patient care by following the best scientific evidence.To provide complete data for quality benchmarking projects, audits and research studies.
To establish a platform to allow long-term follow-up of these infants at a national level. The Registry would facilitate long-term follow-up of a large cohort of twins whose pregnancies were complicated, whether or not they had antenatal intervention.

Mothers Working to Prevent Early Stillbirth Study 20-28
The study’s principal objectives are:
- To conduct structured interviews with bereaved mothers/parents or pregnant people to gain a deeper understanding of potentially modifiable risk factors which affect their baby’s risk of early stillbirth
- To describe associations of potentially modifiable risk factors for early stillbirth according to different causes of early stillbirth.

Tommy’s Respire is funded by Tommy’s, the baby charity, and is being carried out by The Maternal and Fetal Health Research Centre (MFHRC) at St Mary’s Hospital, Manchester University NHS Foundation Trust.
What would my involvement be?
What would I be asked to do if I took part?
Women are asked to participate in different ways; at Leighton Hospital the study only involves a questionnaire. Taking part in The Tommy’s Project may involve:
- Providing information on age, ethnicity, history of any present or previous medical conditions experienced during pregnancy, mode of delivery, birth outcomes, etc. This will help us understand your medical background, any predispositions to developing morbidities in the present or subsequent pregnancies and minimizing risks.
- Providing information on indoor and ambient pollution exposure during pregnancy via questionnaire. Exposure to air pollution can be related to a higher risk of pregnancy-related complications in the parent. Information from this questionnaire can help us establish a link between pollution exposure and unfavourable pregnancy outcomes such as preterm labour and low birth weight amongst others.

PREterm birth genomic investigation using whole Genome sequencing.
Using whole genome sequencing (WGS), the study aims to identify genetic determinants which lead to spontaneous preterm birth (sPTB).
Approximately 10% of global pregnancies are affected by sPTB, this amounts to 53,000 cases in the UK. It is the leading cause of infant mortality globally. A fuller understanding of genetic/biological factors that predispose to PTB will assist in developing strategies to reduce the impact of PTB on the health of affected mothers and children.
PARTICIPANTS
Biological parents (over 18 years old) who had a preterm birth (between 16+0 and 36+6 weeks of gestation) in current or previous pregnancies and children (under 5 years old) born preterm with various ethnic backgrounds across England.

Primary aim: Our NIHR funded pragmatic national trial will compare the effects of ferrous sulphate iron supplementation vs placebo, in non-anaemic pregnant women, on pregnancy outcomes including preterm birth, stillbirth, neonatal death or a small for gestational age birth. We know there is an association between iron deficiency anaemia and adverse outcomes including maternal mortality, haemorrhage, preterm birth, stillbirth, and infant neurocognitive development in the early years of life, but we don’t know how best to prevent the anaemia.
Participants
The study aims to recruit 11,020 non-anaemic pregnant women from participating maternity units over 18 months. We estimate that around 90% of women attending the units for their first visit will be eligible.
Video 1: What is the PANDA study?
https://www.youtube.com/watch?v=4DpYzsVJ1cQ
Video 2: Facts and Myths Around Iron in Pregnancy
https://www.youtube.com/watch?v=IkeWOWmsiUw
Patient Information sheet
https://nhsbtdbe.blob.core.windows.net/umbraco-assets-corp/34387/panda-pis-v12-30jan2024-clean.pdf
Katrina Humphreys is the Physiotherapy Research Facilitator for CCICP MSK studies. She works half time in research and half time in physiotherapy – where her main time is spent as a Vestibular Specialist Physiotherapist. Katrina can be contacted at katrina.
The studies active in CCICP are:
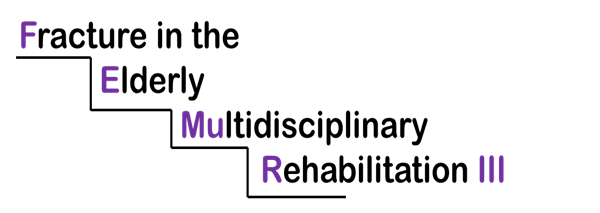
FEMUR 111
This is a Fractured Neck of Femur study – consenting adults over the age of 60 that are admitted to Leighton Hospital with a fractured neck of femur and are then taken to surgery for a repair.
The eligible patients are identified and then asked if they would mind a study team member contacting them when they are discharged home.
If they consent to the study they are then randomised to either the “usual care” arm OR the “enhanced care” arm.
This enhanced care involves 6 visits to the patient’s home to provide physiotherapy rehabilitation over a period of 17 weeks.

PROP-OA
This is a study consenting patients over the age of 45 with osteoarthritis of the knee. This is looking at whether good quality advice and exercises OR that PLUS bracing is more effective for osteoarthritis of the knee.
These patients will be attending the physiotherapy department for an assessment by a physiotherapist and then possibly an X-Ray if needed. Once they are deemed eligible they are booked for a consent and randomisation slot and a treatment time.
These patients then receive their intervention and if they are in the bracing arm of the study they will then have another appointment for a brace check.
A second site has also been opened at Victoria Infirmary.
Medical and Emergency Care (MEC) Research Team
The MEC research team delivers both commercial and non-commercial research for Respiratory medicine, Rheumatology, Cardiovascular disease, Gastroenterology, Anaesthetics as well as some Surgical studies. Business as usual research activity was paused locally and nationally in March 2020 due to the pandemic and, in collaboration with a multidisciplinary team, MEC have led and delivered the Urgent Public Health Research portfolio. A tentative restart of business-as-usual research has now commenced.
Current Studies:

PI Dr J Majumdar; Lead CRN Claire Gabriel
This randomised control trial is looking at different treatments for Covid which currently include:
- High dose Corticosteroids
- Sotrovimab
- Oseltamivir (an antiviral treatment)
- Baloxavir (an antiviral treatment)
- low dose corticosteroids
Treatments that have been proven to work include:
- Dexamethasone reduces risk of death by a fifth in those on oxygen.
- The IL-6 inhibitor Tocilizumab reduces the risk of death by a further seventh
- Neutralising monoclonals can reduce the risk of death by a further fifth in seronegative patients.
Almost 500 patients have been recruited and randomised to the RECOVERY trial at MCHT.
Open to recruitment.

GenOMICC studies the genes (DNA) of people who become critically ill, or meet certain criteria, to better understand why some people become sicker than others and, potentially, to discover new ways of treating patients. More info can be found at www.
Open to recruitment.

The Metoclopramide for Avoiding Pneumonia after Stroke (MAPS-2) Trial
Pneumonia is a major cause of death after stroke and delays recovery in survivors. It is likely to occur in stroke patients who have problems swallowing. The purpose of this study is to test whether the drug metoclopramide can prevent death and pneumonia in patients who have had a stroke.
Open to recruitment.
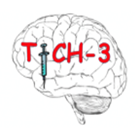
Tranexamic acid for Hyperacute Spontaneous Intracerebral Haemorrhage (TICH-3)
The purpose of this study is to see if the drug tranexamic acid reduced the bleeding in this type of stroke and if it may reduce the severity of injury caused by the stroke.
Open to recruitment.

Pharyngeal Electrical stimulation for Acute Stroke dysphagia Trial (PhEAST)
The purpose of this study is to find out whether a regular course of treatment of Pharyngeal Electrical Stimulation (PES), stimulation of nerves in the throat that may have been damaged by a stroke, can help recovery back to eating and drinking by mouth.
Open to recruitment.

The aim of this randomised study is to investigate which treatments are best for patients admitted to the ICU with flu or pneumonia.
The treatments for flu are:
- oseltamivir
The treatments for other pneumonia are:
- IV Hydrocortisone
- Dexamethasone
- antibiotics which may help reduce inflammation.

The ABBRUPT trial compares two of the effective and routinely used treatments for atrial fibrillation – amiodarone and beta blocker. However, clinicians do not yet know which treatment works best and ABBRUPT aims to discover which one (if any) works best for new onset atrial fibrillation.

A randomised research study to find out if awake prone positioning (laying on your front) is better than standard care for patients with respiratory failure.

A randomised, placebo-controlled feasibility trial evaluating remote ischaemic conditioning (RIC) after acute ischaemic stroke. Evidence from experiments and other conditions suggests that interrupting the blood supply to the arms (for example, by inflating blood pressure cuffs) for brief episodes may help protect the brain from further damage.
Trials no longer recruiting but still collecting follow-up data:
L-Harp
Serena
Ophthalmology Research
Leighton Hospital’s Ophthalmology Department is actively involved in research. The two studies currently open are important in helping us to better understand how to diagnose, treat, cure or prevent age-related macular degeneration. In the coming months we will be expanding our portfolio to include areas of dry eye, glaucoma and cataract research studies.
If you would like to know more about getting involved in any clinical research in this area please contact Advanced Nurse Practitioner Lisa Rudy Fitzgerald on bleep 3451 (accessed via main switchboard 01270 255141).